 Gray Medical
Gray Medical
 Gray Medical
Gray Medical
In the modern clinical landscape, Patient Monitoring Solutions have transcended mere data display. The shift towards the Internet of Medical Things (IoMT) has transformed monitoring devices into predictive analytical hubs. From continuous bedside hemodynamic monitoring to remote patient management (RPM) for chronic disease oversight, China's manufacturing sector has evolved from component assembly to full-cycle medical technology innovation.
Tianjin Gray Medical Technology Co., Ltd., along with the broader Chinese medical manufacturing ecosystem, demonstrates profound supply chain resilience. By integrating vertical manufacturing—from raw material sourcing for medical consumables to high-precision CNC machining for orthopedic implants—we ensure consistent quality control. This end-to-end efficiency is critical in the post-pandemic era, where global medical institutions require not just "fast" delivery, but uninterrupted, ISO-certified reliability. Our facilities utilize automated robotics and AI-driven quality assurance to maintain compliance with international standards such as CE and FDA benchmarks.
The future of patient monitoring lies in Clinical Decision Support Systems (CDSS). We are actively incorporating AI algorithms into our diagnostic equipment to reduce "alarm fatigue" for nurses and provide early warnings for sepsis or cardiac events. Looking ahead, our R&D roadmap focuses on:
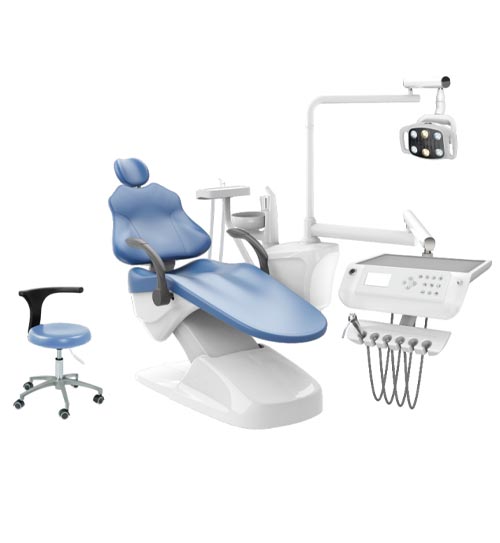
Founded in 2012 in Tianjin, our journey reflects a decade of commitment to clinical excellence. We provide comprehensive solutions covering operating room instruments, rehabilitation devices, and diagnostic tools. Our philosophy is rooted in Evidence-Based Design—creating products that not only meet medical requirements but actively simplify the workflow for healthcare professionals.
Our commitment extends to localized support. Whether you are a hospital in Europe, an SME clinic in Southeast Asia, or a distributor in the Americas, our service system—including 24/7 technical support and professional training—ensures our equipment performs at peak operational capacity throughout its lifecycle.
We adhere to strict GMP and ISO standards. Each product undergoes rigorous testing—from raw material screening to clinical evaluation—ensuring our exports meet the regulatory demands of global markets including the EU and FDA jurisdictions.
Yes. Our "factory-to-client" model allows for modular customization. We work closely with clinical teams to adjust specifications for surgical tools, diagnostic equipment, and patient monitoring systems to fit specific environmental constraints.
Our automated production lines and deep supply chain integration enable us to maintain optimized inventory levels, significantly reducing lead times compared to industry averages without compromising quality assurance.