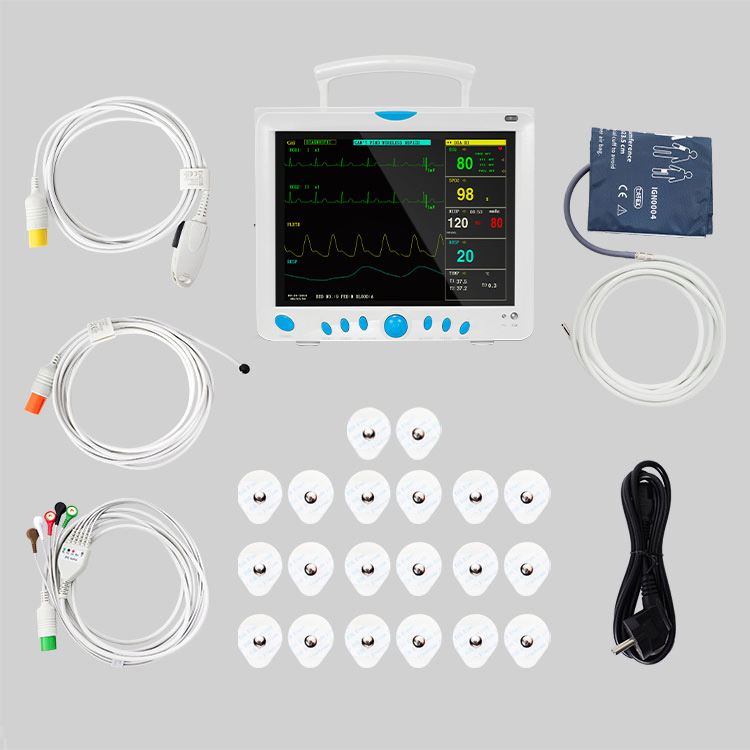
1 / 5
 Gray Medical
Gray Medical










| Customization: | Available |
|---|---|
| Classification: | Physiological Functions of Diagnosis and Monitoring Equipment |
| Type: | Patient Monitor |
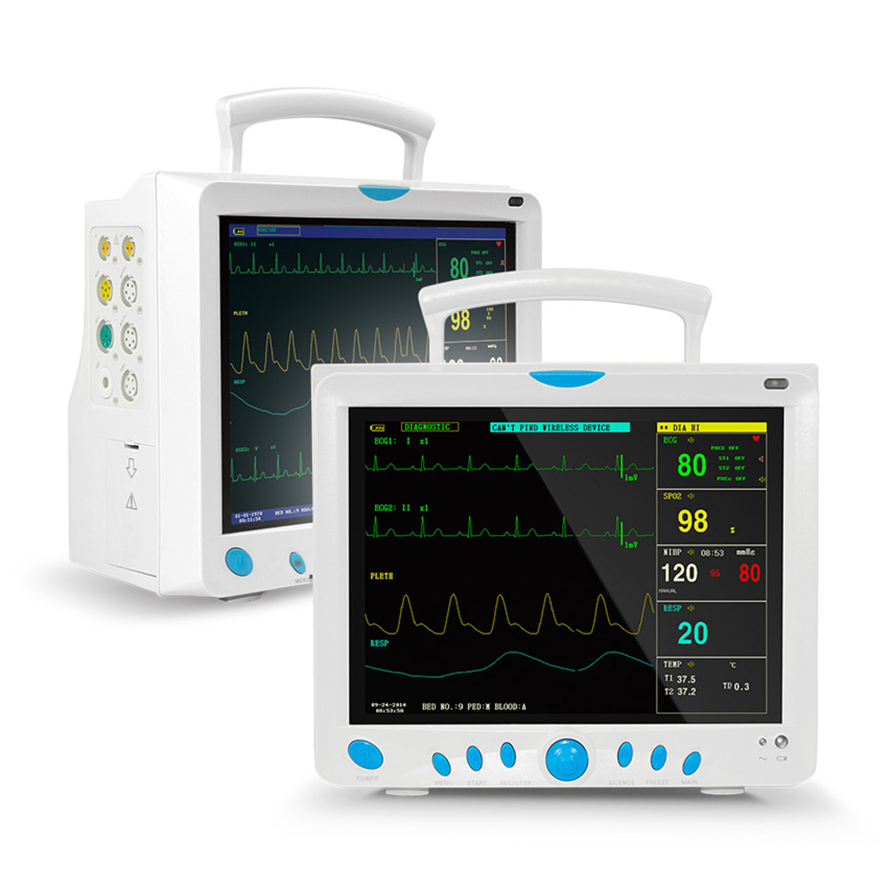
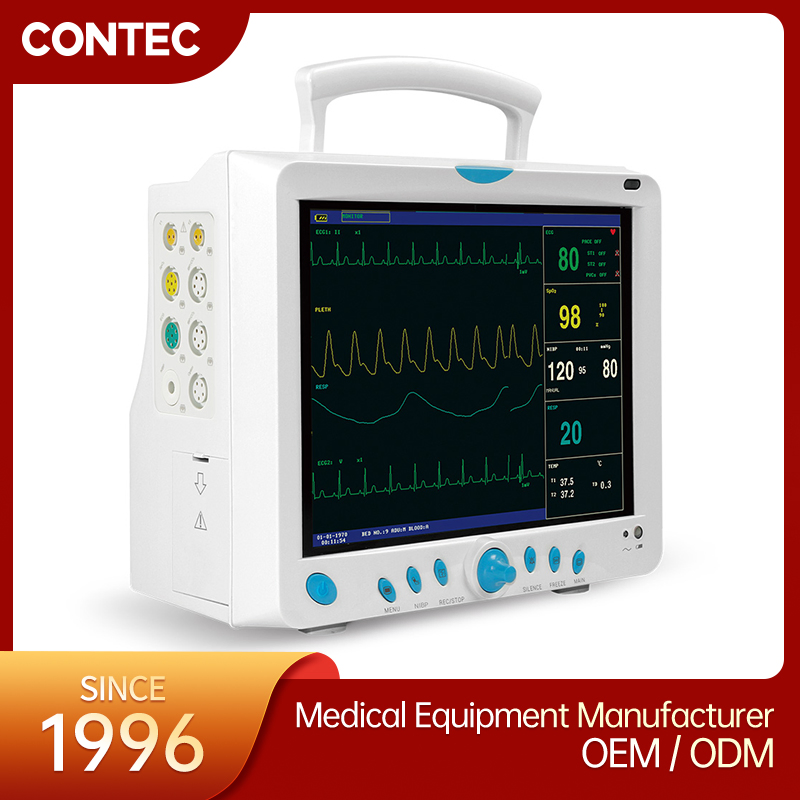
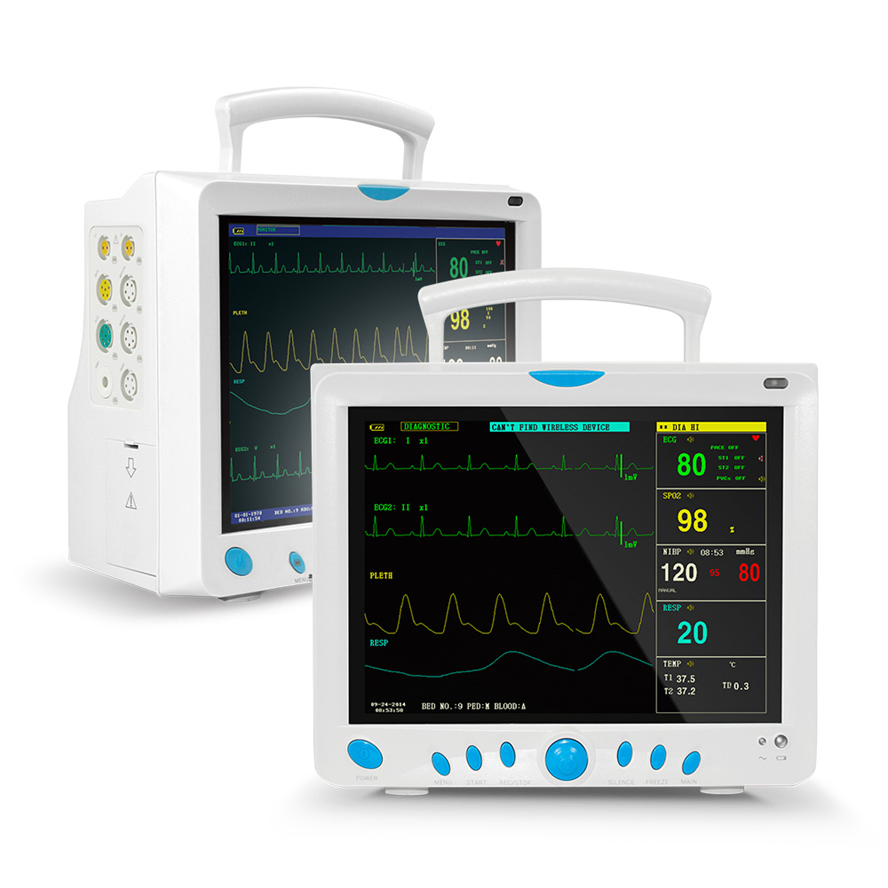
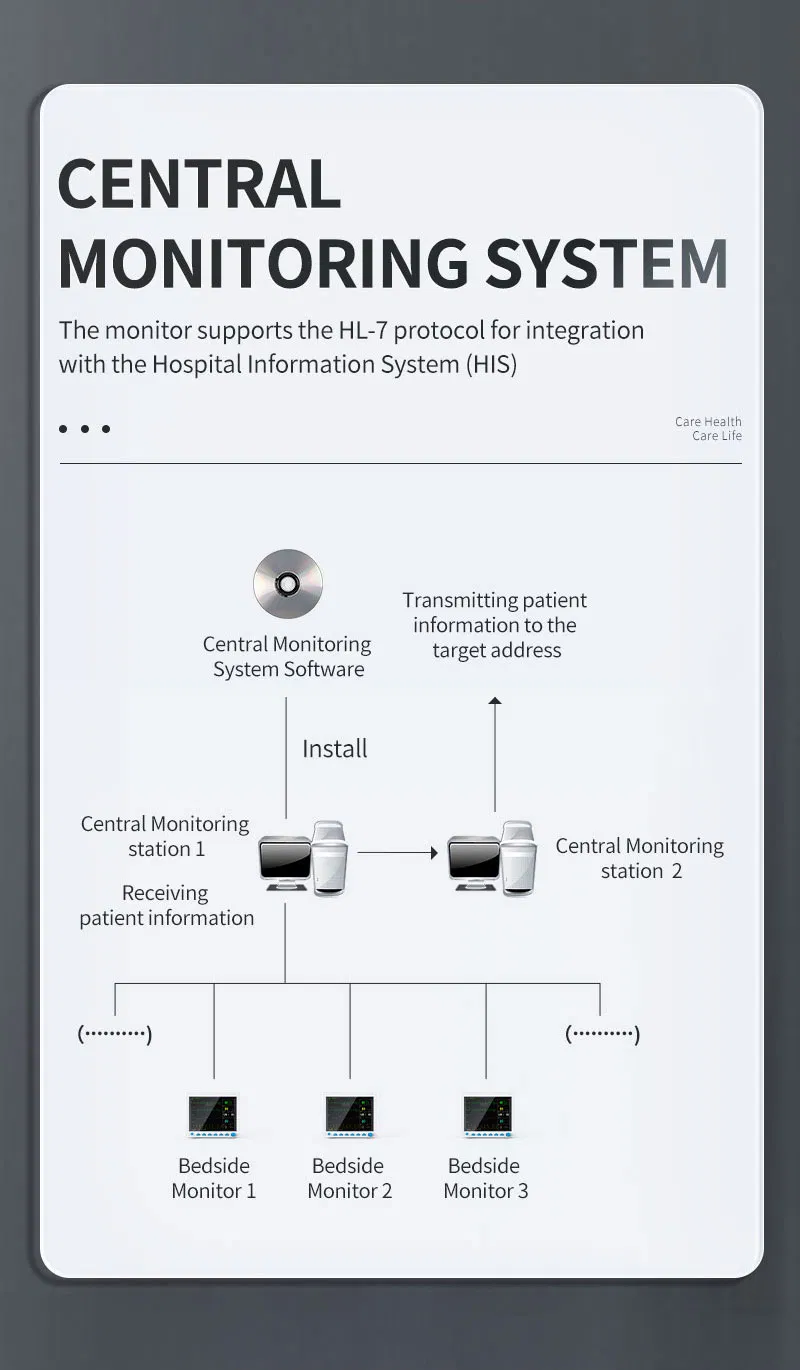
Professional Grade CMS9000 Patient Monitoring System


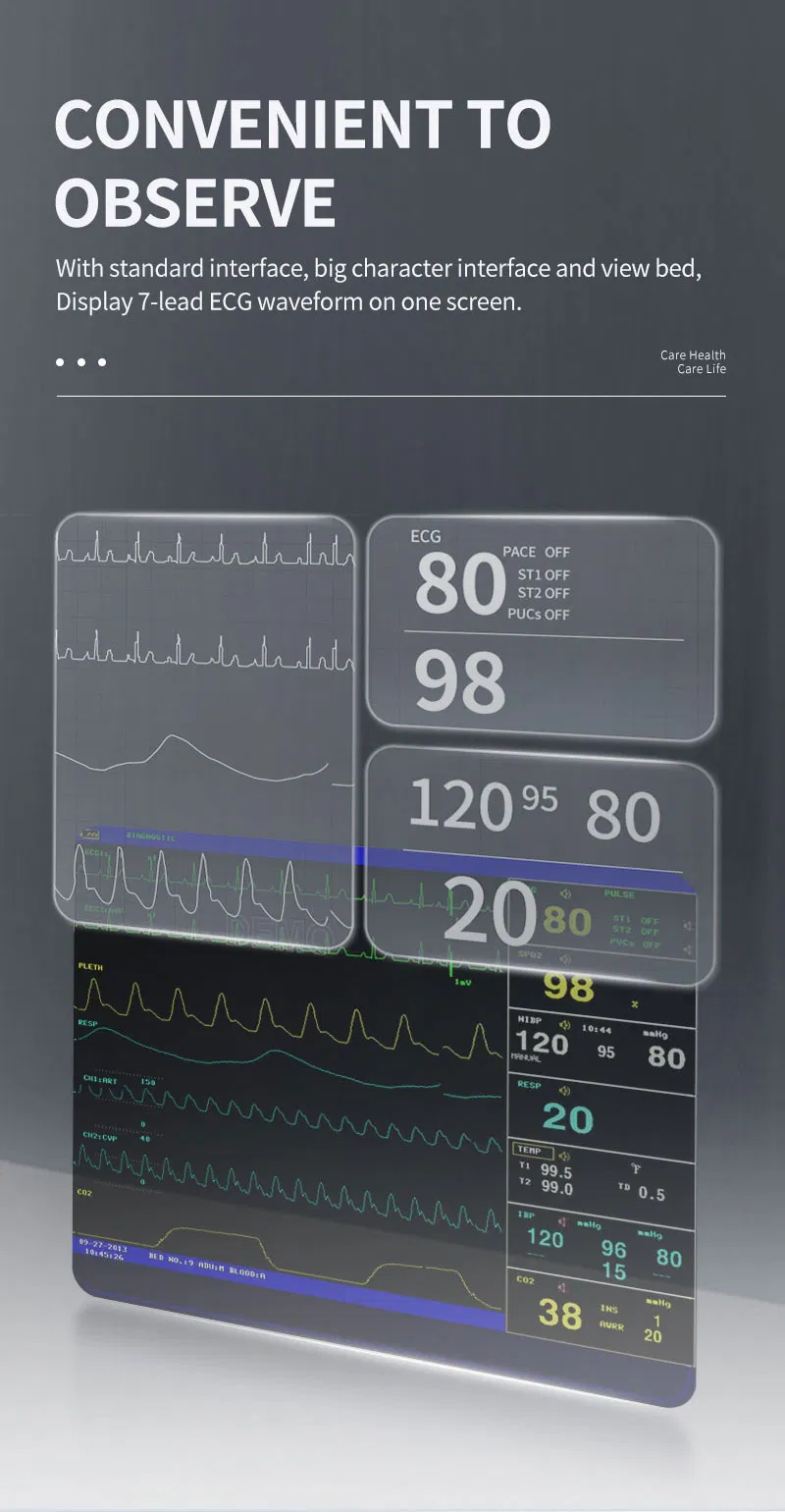
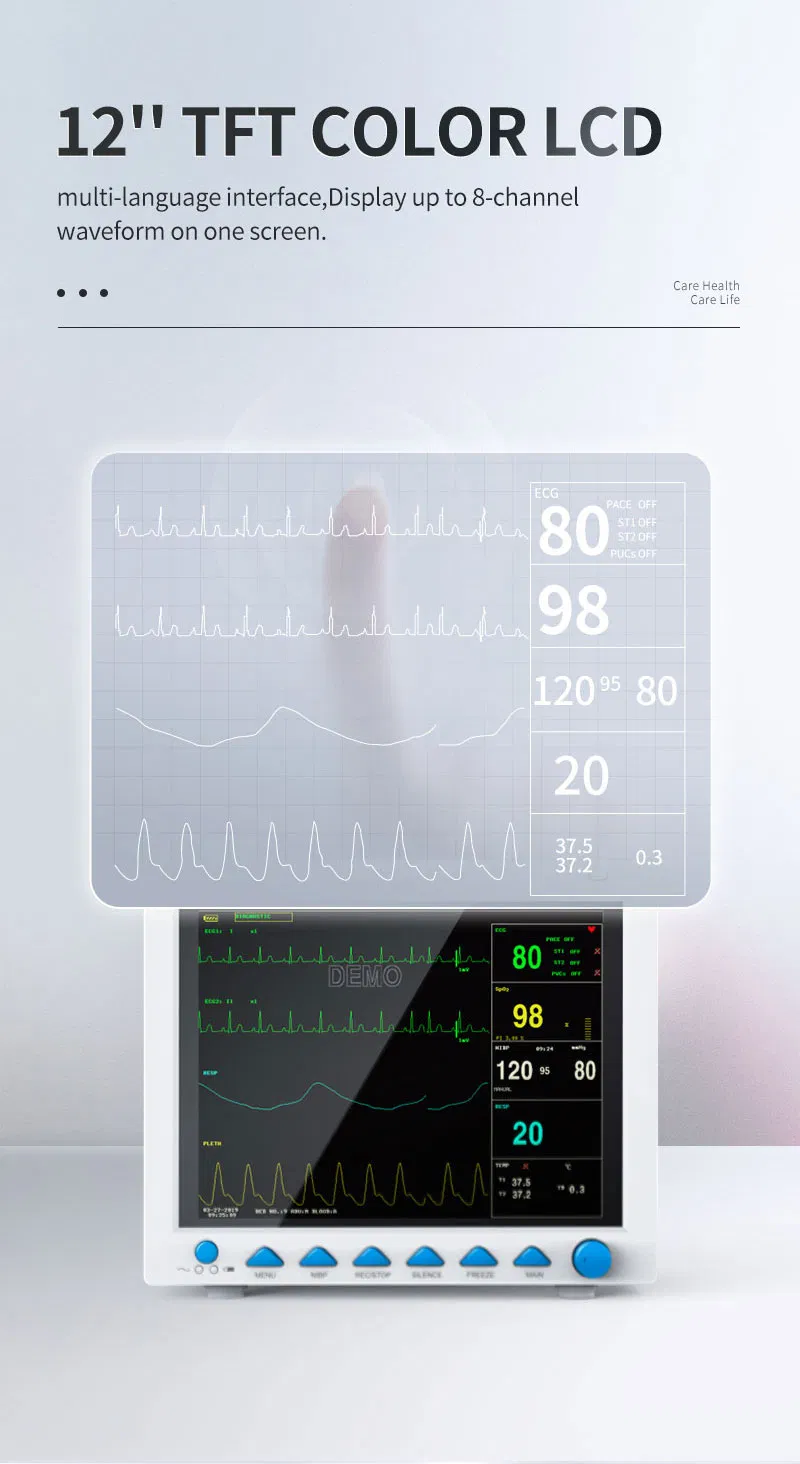

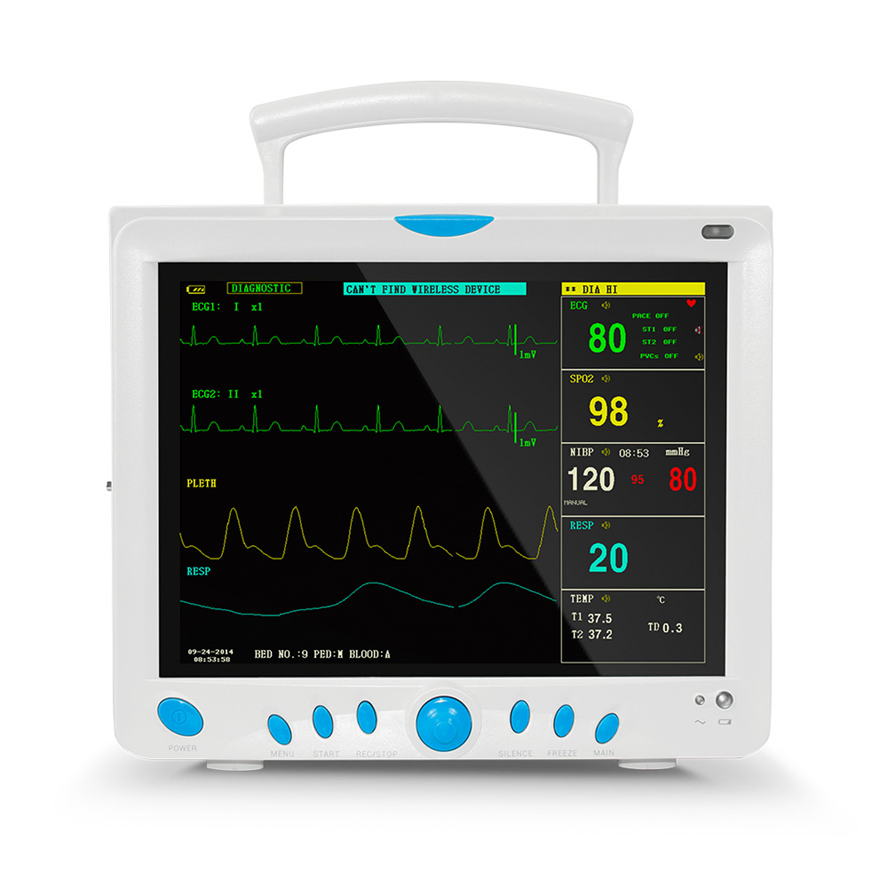
5-lead or 3-lead selectable. Gain selection: 12.5mm/s, 25mm/s, 50mm/s. Accuracy: ±1bpm or ±1%. Features S-T detection and Arrhythmia analysis.
Method: Oscillometry. Operation modes: Manual/Automatic/STAT. Measurement types: Systolic, Diastolic, Mean. Range: 10-270mmHg for adults.
SpO2 Range: 0-100% with ±2% accuracy (70-100%). Respiration Method: RA-LL impedance. Apnea alarm included.
Support for EtCO2 (Side-stream or mainstream), IBP (2 channels), and CO (Thermo-dilution method).







