



 Gray Medical
Gray Medical










| Still deciding? Get samples of $ ! US$ 1.5/Piece Request Sample |
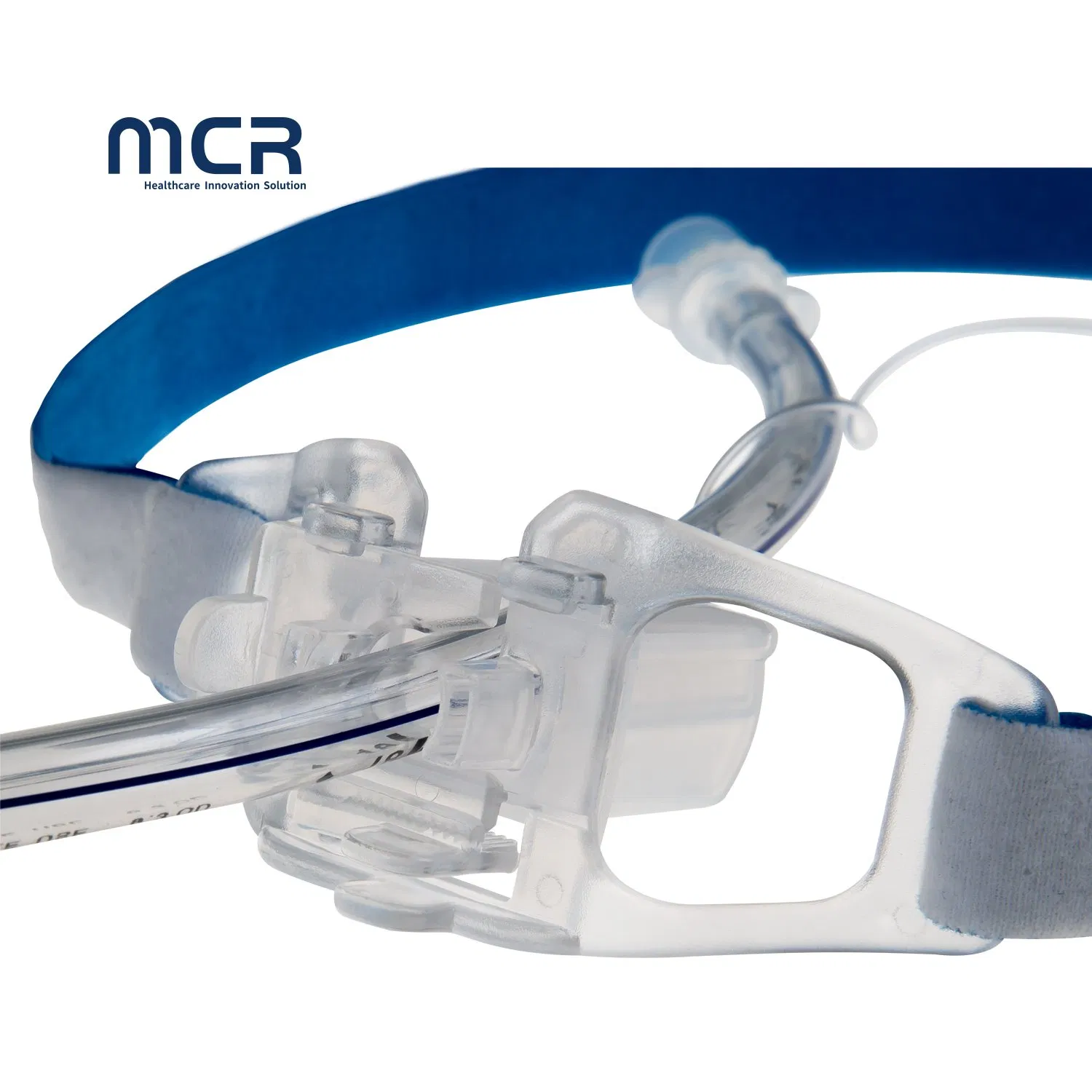
The Tracheal cannula fixation device is designed to ease patient suffering and significantly reduce medical complications associated with intubation. It is primarily used for securing endotracheal intubation to prevent loosening or accidental displacement.
This ICU-grade device securely surrounds the back of the head and passes beneath the ears to stabilize both sides of the oral endotracheal tube effectively.










With 16 years of expertise in manufacturing medical intubation products, we have developed mature technology and production processes. Our commitment to innovation is driven by global medical trends, focusing on the visualization of intubation products.
Our current and future goals involve transitioning expensive fiberscopes and endoscopes into disposable, cost-effective solutions to prevent cross-infection, ensuring safety for both medical professionals and patients across urology, gynecology, and gastroenterology departments.