



 Gray Medical
Gray Medical










| Still deciding? Get samples of $ ! US$ 1.45/Piece Request Sample |
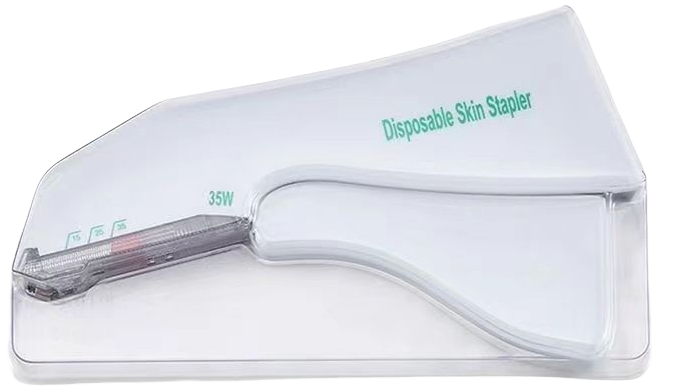
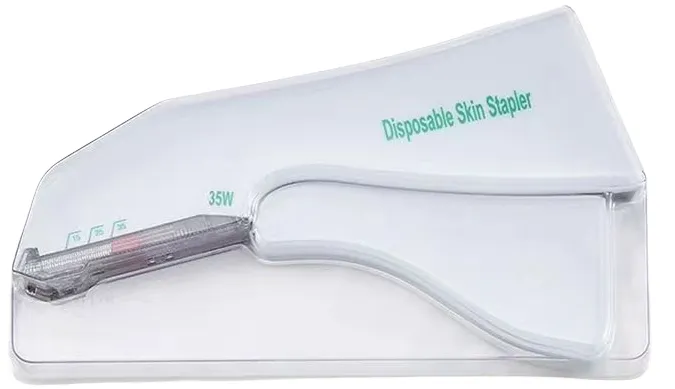
| Model NO. | Surgical Skin Stapler |
| Sterilization | Ethylene Oxide (EO) |
| Quality Guarantee | 5 Years |
| Group | All |
| Logo Printing | Available |
| Product Color | White |
| Packaging | Single Aluminum Bag Packing |
| Carton Size | 46.5X45X22cm |
| Transport Package | 80PCS/CTN |
| Specifications | 15W, 25W, 35W, 15R, 25R, 35R, 15H, 25H, 35H |
| HS Code | 9018322000 |
| Production Capacity | 10,000 pieces/Day |
| Type | Surgical Skin Stapler |
| Colour | White |
| Model | 15W, 25W, 35W, 15R, 25R, 35R, 15H, 25H, 35H |
| Sterilization | Ethylene Oxide |
| Certificates | FDA, FSC |




We specialize in the production of medical consumables, established with a registered capital of 30 million. The main products are absorbable surgical suture with needle (Plain / Chromic Catgut, PGA, PDO, PGLA, PGCL, etc.), non-absorbable surgical suture with needle (Silk, Nylon, Polyester, Polypropylene etc.), suture materials, cassette sutures and surgical needle (300SS and 400SS Series), lifting threads for beauty, medical facial mask, orthopedic casting tapes & splints, plaster, IV cannula, polypropylene meshes and absorbable bone nails.
We have approved ISO13485 international quality system certificate, CE and FDA certificates, and obtained the "Medical Device Manufacturing Enterprise License" and "Medical Device Registration Certificate". We operate one of the largest catgut plants and serve as a core enterprise for surgical absorbable sutures. The products are exported to Asia, Europe, Latin America, Africa, Middle East and more than 70 countries and regions worldwide.


