

1 / 5
 Gray Medical
Gray Medical










| Still deciding? Get samples of $ ! US$ 1/Piece Request Sample |
For professional and in vitro diagnostic use only.
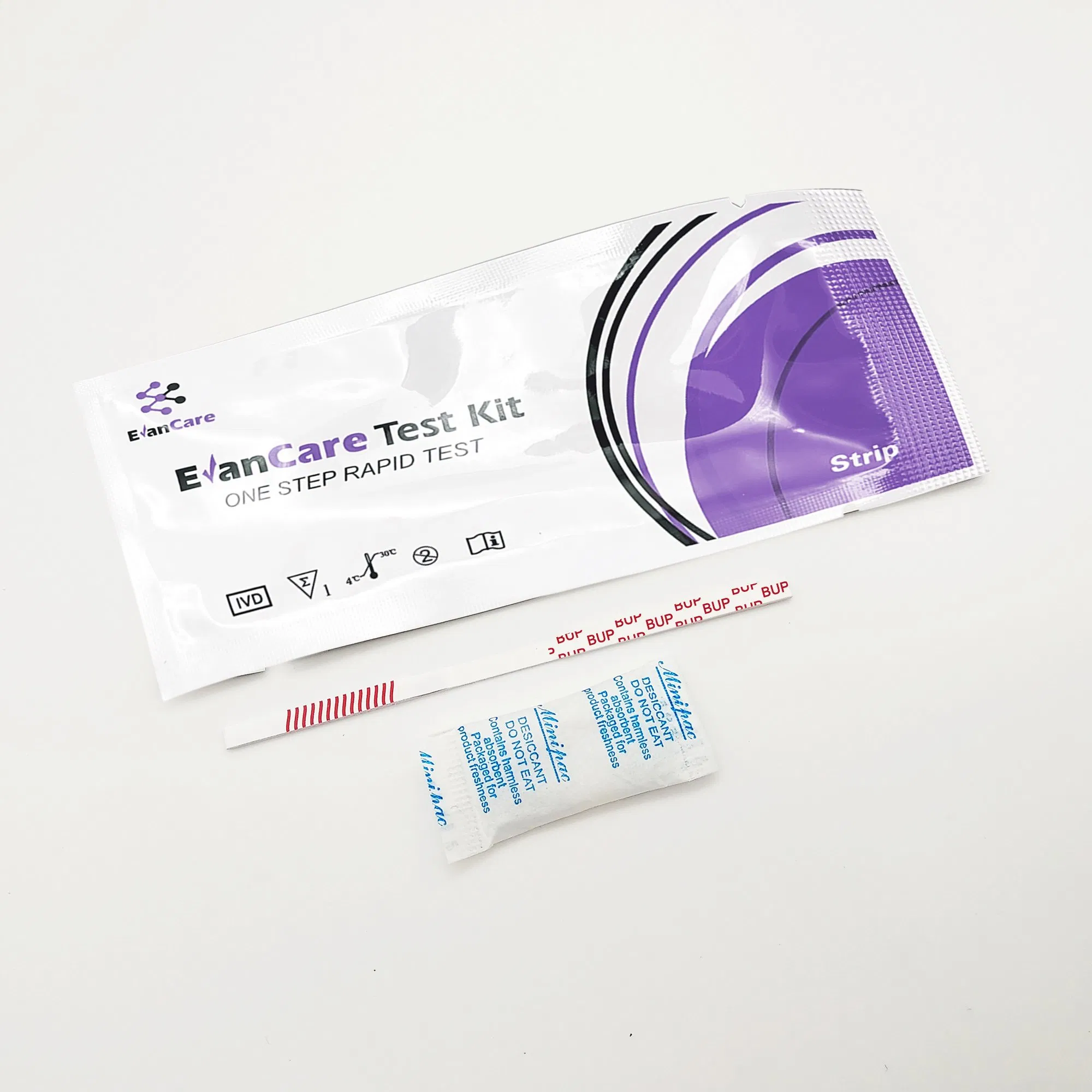
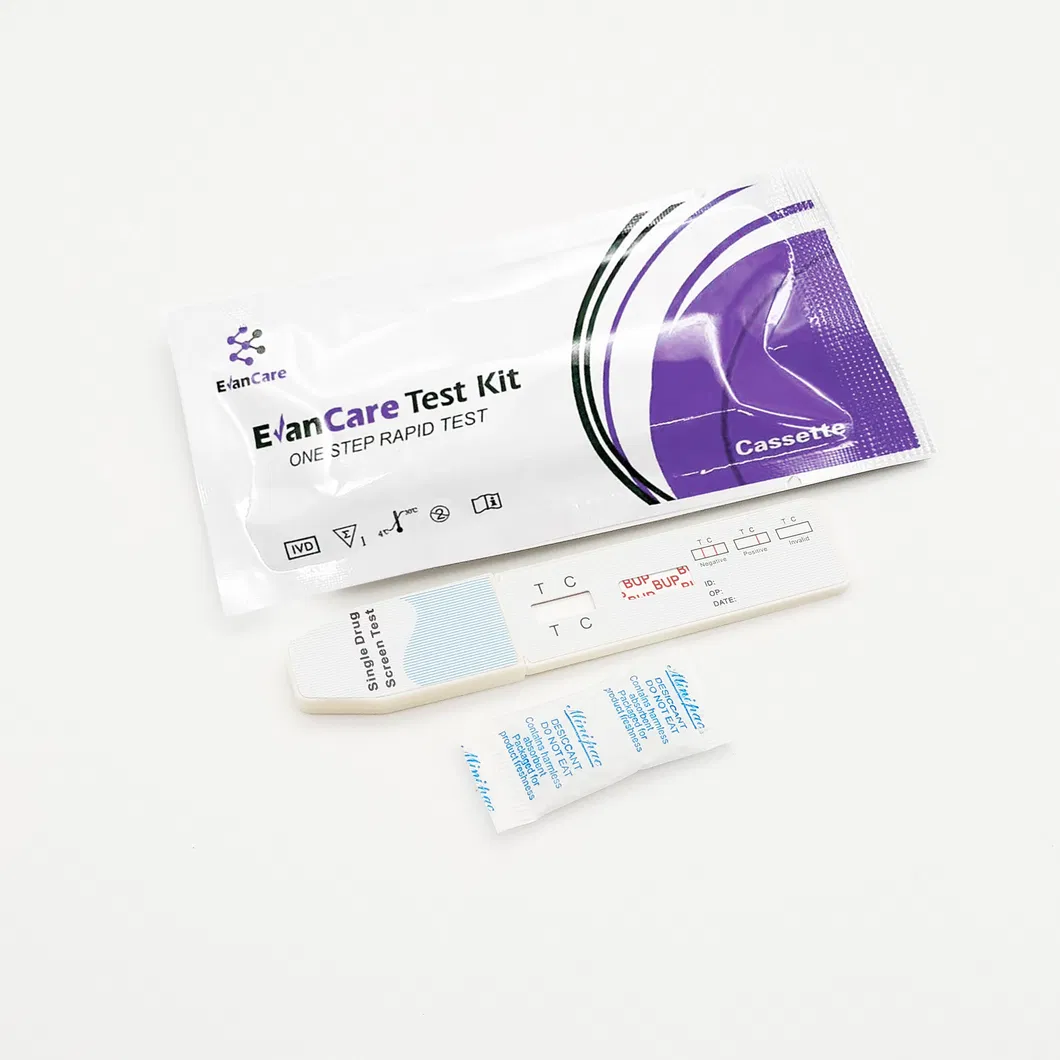
[INTENDED USE]: The Rapid Test Panel (Urine) is a rapid visual immunoassay for the qualitative, presumptive detection of substances in human urine specimens at specific cut-off concentrations.
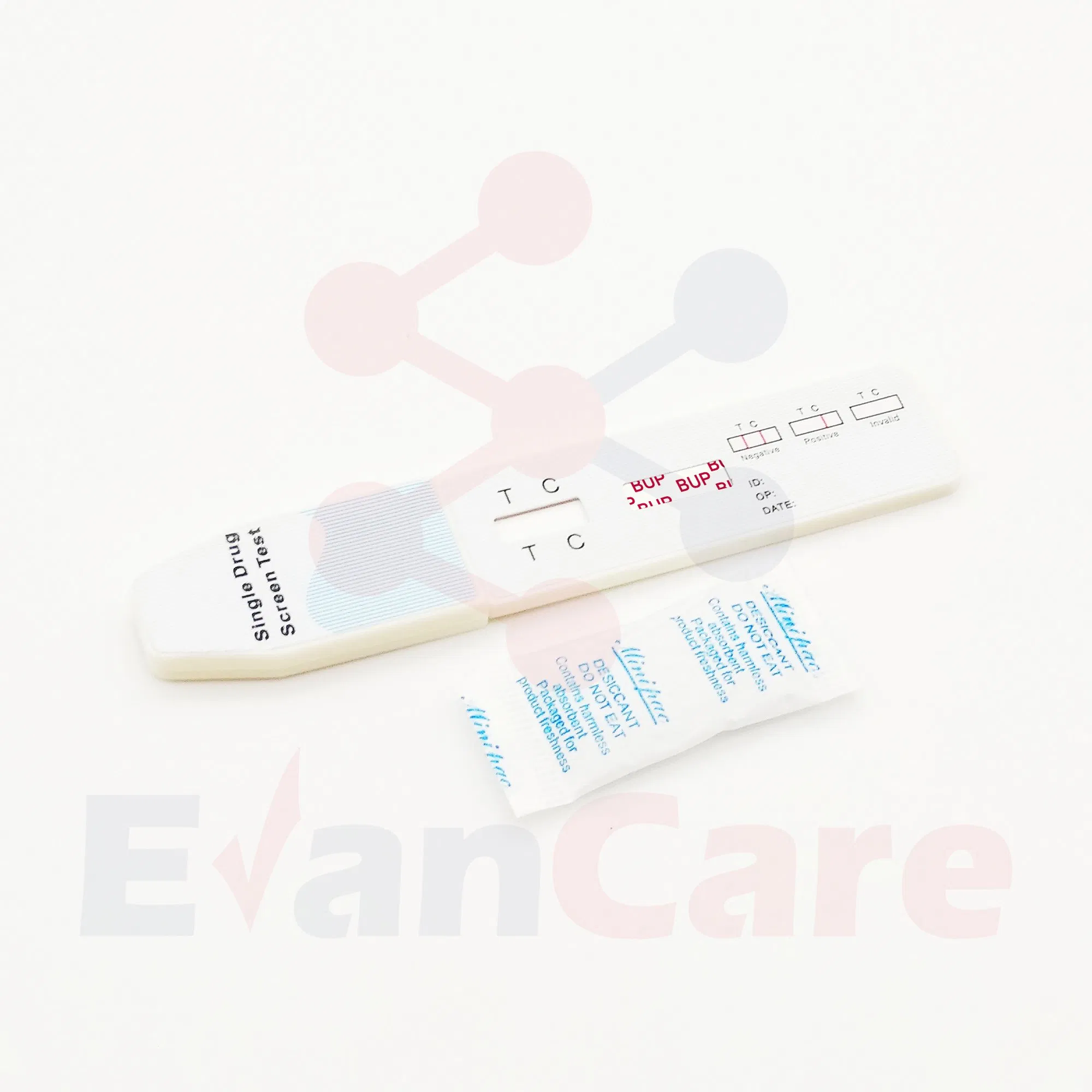
[SUMMARY]: This test detects Buprenorphine through visual interpretation of color development. If there are insufficient drug molecules in the specimen, a colored band forms at the test region (negative). If drug molecules are present above the cut-off, no band appears (positive). A control band (C) always appears to indicate proper volume and procedure.

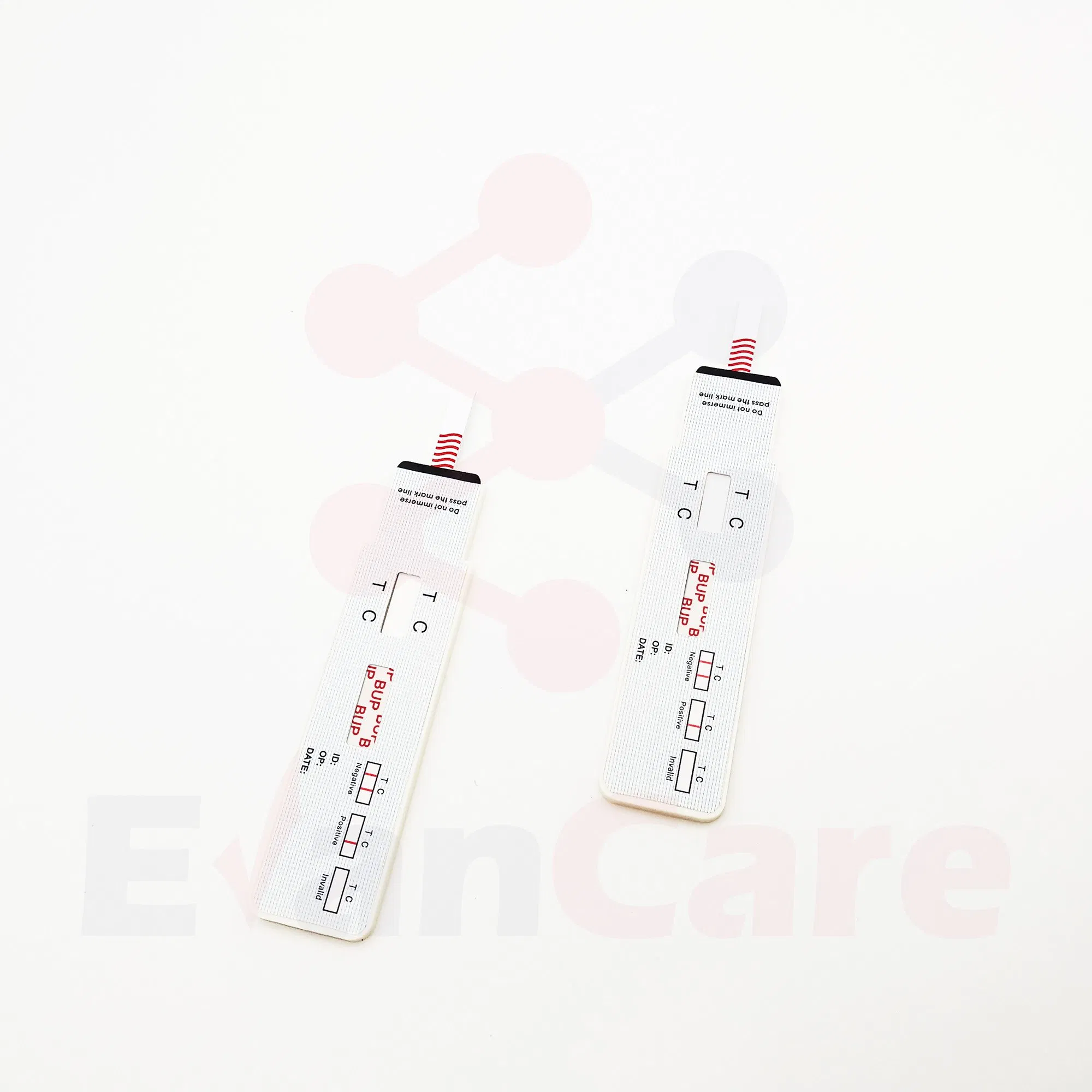
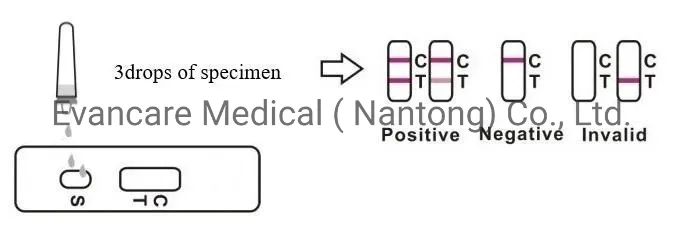
| Positive | One colored line appears in the control region (C). No line appears in the test region (T). |
| Negative | Two lines appear. One colored line in the control region (C) and another in the test region (T). |
| Invalid | Control line fails to appear. Review the procedure and repeat with a new test cassette. |
| Contents | 1 Test Cassette, 1 Desiccant per bag; 25 units per box |
| Format / Size | Cassette: 2.5mm / 3.0mm / 4.0mm |
| Methodology | Sandwich Colloidal Gold |
| Specimen | Urine |
| Expiry Date | 3 Years |
| Accuracy | > 99.99% |
| OEM Service | Available |






