




 Gray Medical
Gray Medical










| Still deciding? Get samples of $ ! US$ 999/Piece Request Sample |
| Model NO. | HD 12 |
| Certification | CE, FDA, MSDS, ISO13485 |
| Group | for All Age Groups |
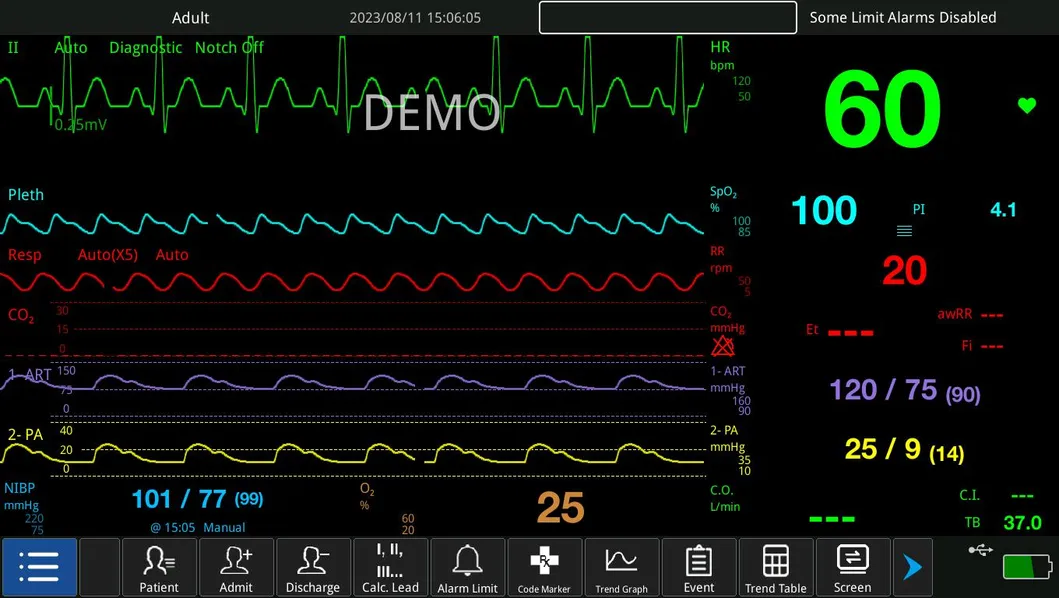
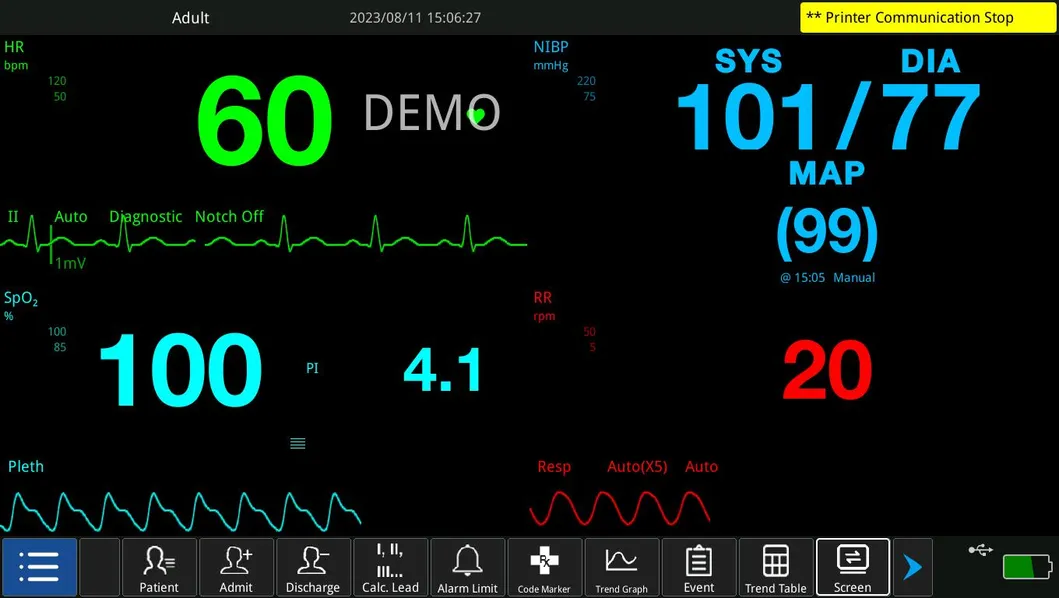
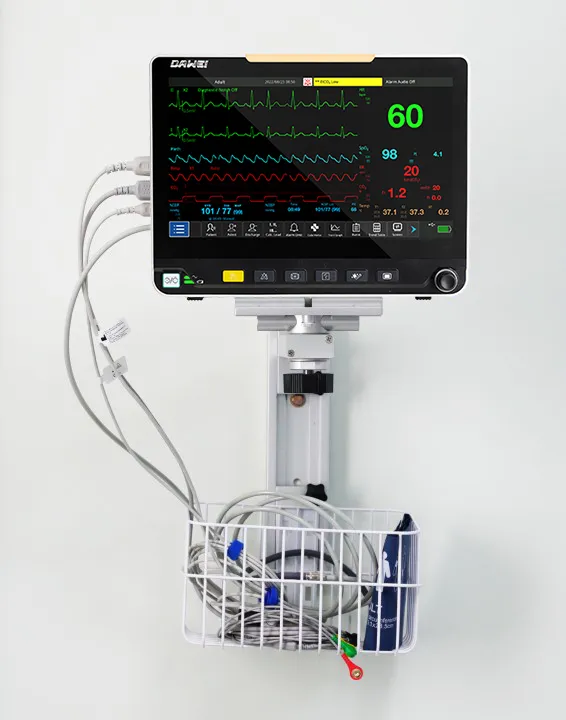
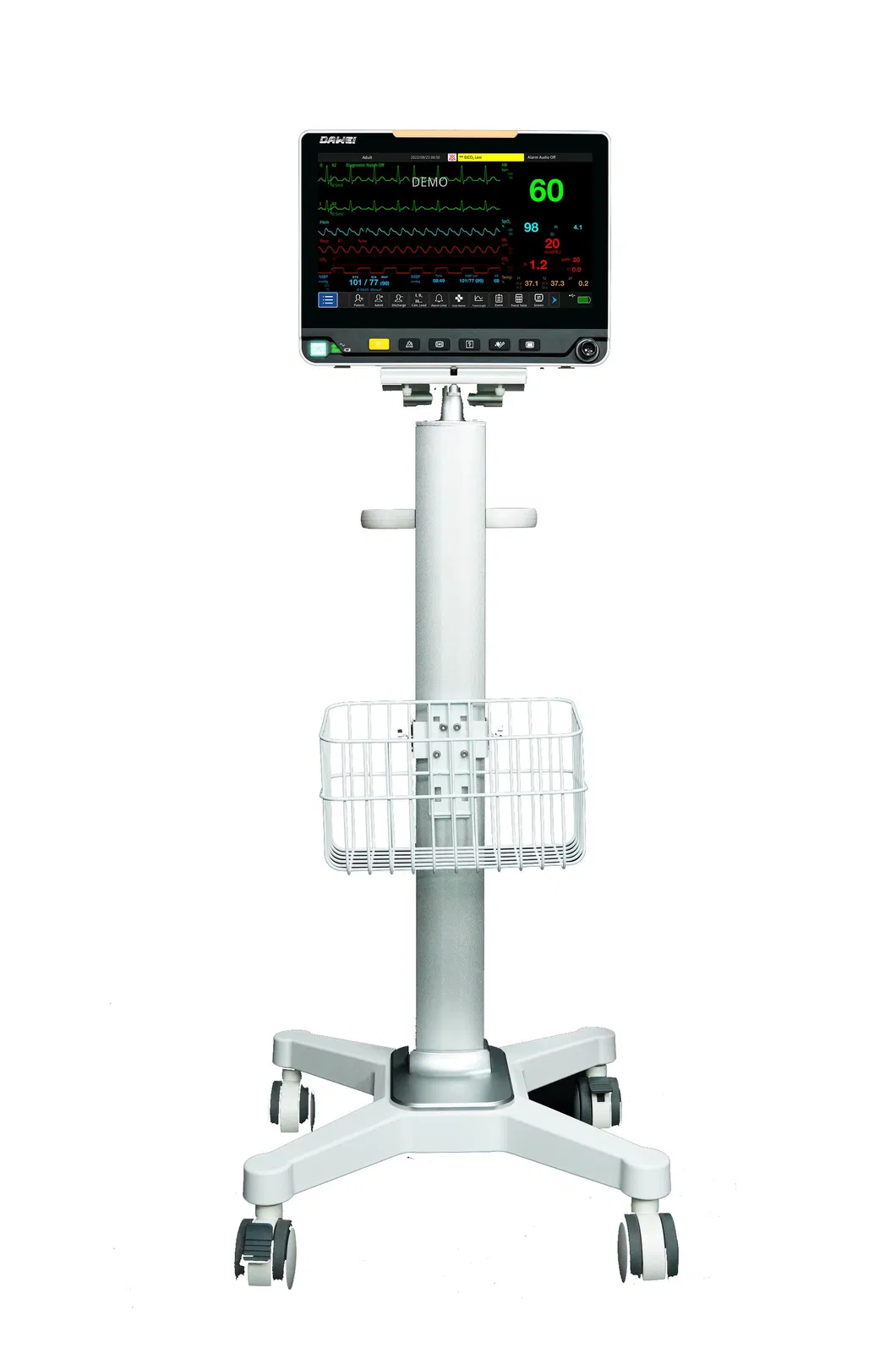
| Product | Multi-Parameter Monitor |
| Modules | ECG, NIBP, Hr, Temp, SpO2 |
| Display | 13.3 Inch |
| Languages | 4 Languages |
| Available | ICU Room |
| Transport Package | Export-Dedicated Paperboard Boxes |
| Specification | 390*290*340(L*D*H)mm |
| Production Capacity | 20000/Year |
| Standard Configuration | Optional Modules |
| ECG, RESP, NIBP, SpO2, PR, TEMP | IBP, CO, ICG, EtCO2, AG, RM, BIS |


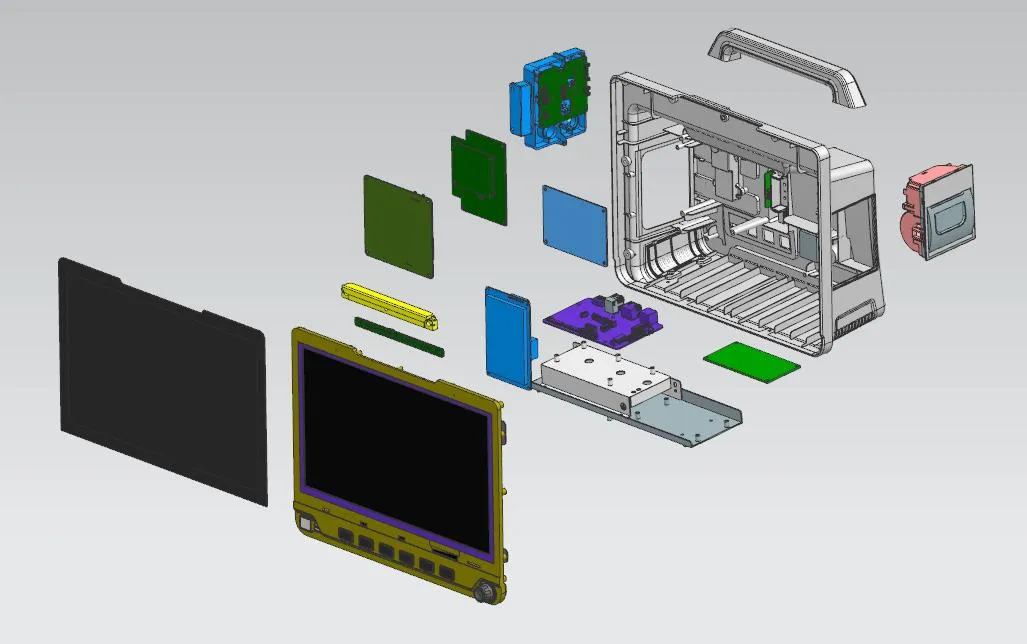
We provide tailored OEM/CKD Services for your unique needs to ensure the equipment fits your specific medical environment.



ISO 13485 Certification
ISO 13485 certification is an esteemed international standard designed specifically for the medical device industry. This underscores the critical importance of a meticulous quality management system, ensuring products consistently adhere to the exacting standards of the medical industry.
Our production processes undergo exhaustive scrutiny to ensure product uniformity and compliance with all regulatory standards. We actively integrate pioneering technologies to enhance product performance and reliability.
Established as a beacon of excellence in the domestic medical device industry, we have tirelessly pursued innovative advancements in ultrasound imaging, digital electrocardiography, and digital radiology since 2006.
The product portfolio boasts over 50 models spanning major categories, including color Doppler ultrasound diagnostic systems, advanced ECG machines, sophisticated patient monitors, and digital radiography equipment. All products are certified with ISO 13485 and European CE quality certification.



Our after-sales service team comprises seasoned technical experts adept at providing rapid responses. We offer remote technical support through phone, email, or specialized remote assistance tools. When necessary, we offer on-site repair services to ensure equipment is swiftly restored to optimal functionality.
